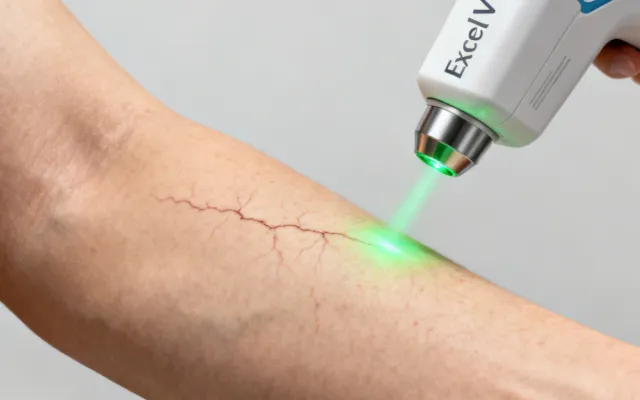
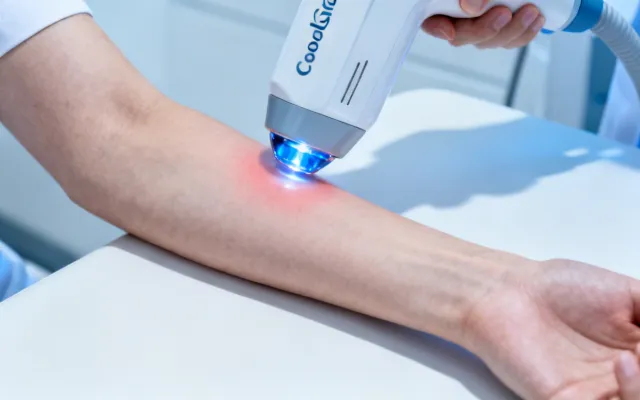
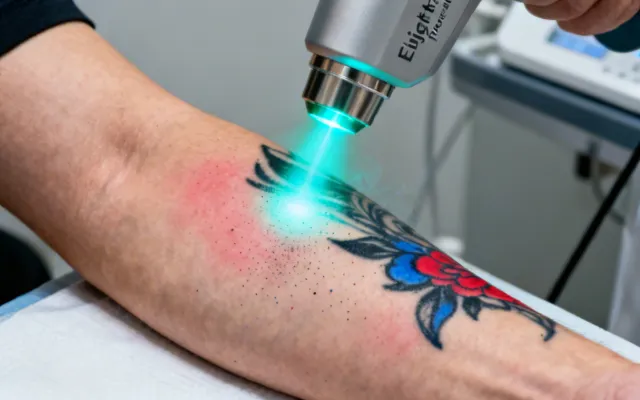
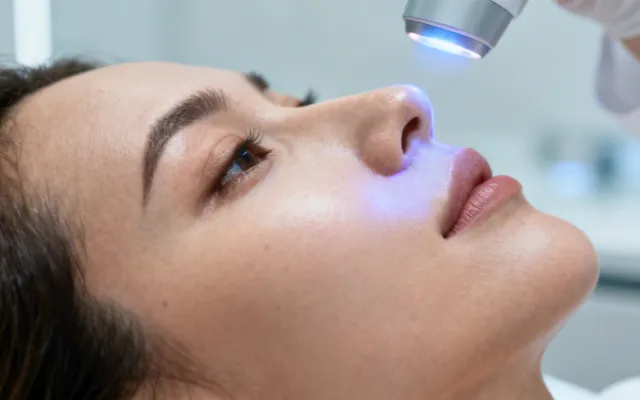
Cutera Laser Genesis & Clinical Treatment Solutions
Evidence-based treatment protocols matched to specific dermatologic and aesthetic indications. Each protocol is developed from published clinical data and refined through our application laboratory's experience with over 15,000 installed systems worldwide.